Abstract
Background: For multiple myeloma (MM) and AL amyloidosis (AL) patients undergoing autologous hematopoietic stem cell transplant (AHCT), high-dose melphalan is given at a fixed dose based on body surface area with five-fold inter-patient variability in exposure, as measured by area under the curve (AUC). Patients with higher than the median AUC (12.85mg/L*h) have prolonged survival, but increased toxicity (Shaw et al., BBMT 2012). Pharmacokinetically (PK)-directed dosing has the potential to achieve an optimal concentration in each patient. We prospectively evaluated the feasibility of PK-directed dosing of propylene glycol free melphalan (Evomela®, PGF-MEL), a more stable and possibly less toxic intravenous formulation of melphalan, to personalize therapy for maximum efficacy and tolerability.
Methods: MM and AL patients age 18-75 received a 10mg/m2 test dose of PGF-MEL within 14 days of AHCT. Serum samples were drawn at 5, 15, 30, 40, 75, and 150 min after PGF-MEL administration. Liquid chromatography tandem mass spectrometry was used to measure MEL concentrations. AUC PK modeling with Phoenix WinNonlin v6.4 was used to determine a target dose to achieve an AUC of 13.5mg/L*h (+/-1), which was given on day -2 prior to AHCT. PK samples were collected after the target dose at the same time points as after the test dose. AHCT with 3-10 x10^6 CD34+ cells/kg was conducted per institutional guidelines. Patient-reported symptom burden was collected prospectively using the MD Anderson Symptom Inventory - MM (MDASI-MM) on day +1, 6, 11, 30, 60, and 90. Toxicities were collected by chart review through day +100 after AHCT and compared using either a T-test or Fisher's exact test between patients above and below AUC 14.5 mg/L*h.
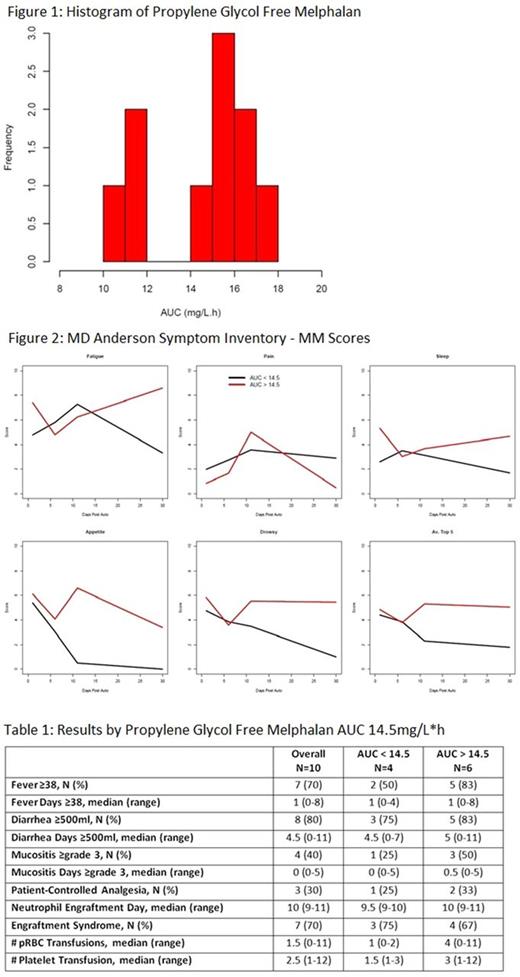
Results: Ten patients (7 MM, 3 AL) with 50% male, median age 67yr (range 53-74), and median body surface area 1.95 (range 1.56-2.35) had a median of 4.5 days (range 1-24) between the test and target doses of PGF-MEL. Target doses were calculated for 90% of patients and 70% received the calculated dose. The reasons for not administering the calculated dose were inability to calculate a test dose AUC (N=1), toxicity after the test dose (N=1), and target dose outside study safety parameters (N=1). Median test and target dose AUCs were 0.655 (range 0.447-0.869) and 15.65mg/L*h (range 10.1-17.2, Figure 1), respectively. No patients receiving the calculated target dose had an AUC within the goal range. MDASI-MM AUCs were calculated for fatigue, pain, sleep, appetite, drowsiness, and the average of these 5 symptoms. Overall, higher scores were seen in patients with MEL AUCs above 14.5mg/L*h compared with below (Figure 2). More patients with higher AUCs had fevers (83 vs 50%), ≥500ml of diarrhea per day (83 vs 75%), grade ≥3 mucositis (50 vs 25%), and need for patient-controlled analgesia (33 vs 25%), but these were not statistically significantly different between the groups (Table 1). Neutrophil engraftment occurred at a median of 10 (range 9-11) days post AHCT in both groups; patients with AUCs above 14.5mg/L*h required more pRBC and platelet transfusions. With a median follow-up of 217 days (range 179-250) in surviving patients, no patients progressed, and one AL patient died of failure to thrive at 134 days post AHCT.
Conclusion: Collection of PK levels and calculation of a target dose was feasible. However, using a 10mg/m2 test dose we did not achieve the target AUC in our patients. This could be due to the small test dose which may have been insufficient to saturate the two-compartment model, rendering the PK analysis inaccurate. Patients with higher AUCs required more transfusions, had more toxicities, and had prolonged patient-reported symptom burden. Due to the small number of patients, these differences were not statistically significant, but suggest there may be an ideal concentration to minimize toxicity and maximize efficacy. Disease response and outcome data continue to be collected. Further study is needed to determine the optimum test dose and schedule, and modifications to the schema are ongoing.
Koehne: Atara: Consultancy, Patents & Royalties. Landau: Janssen: Honoraria; Takeda: Research Funding; Spectrum Pharmaceuticals: Other: Advisory Board, Research Funding; Celgene: Other: Advisory Board; Pfizer: Honoraria; Karyopharm: Consultancy; Amgen: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal